Research
Yokokawa group focuses on the development of microfabricated devices with applications in tissue engineering, disease modeling, stem cell studies, pharmacology, synthetic biology and regenerative medicine. We instigate and promote synergy among various interdisciplinary research projects in our group to deepen our understandings of biological functions at cellular and organ level. The goal is to provide solutions not only to improve the quality of life (QOL) but also to enable treatment and diagnosis of diseases by personalized medicine globally. The group consist of biomedical engineers, mechanical engineers, and biologist, involved in research areas encompassing a broad range of topics from organs-on-a-chip (microphysiological systems) to biophysics of motor proteins.
Kidney and bladder
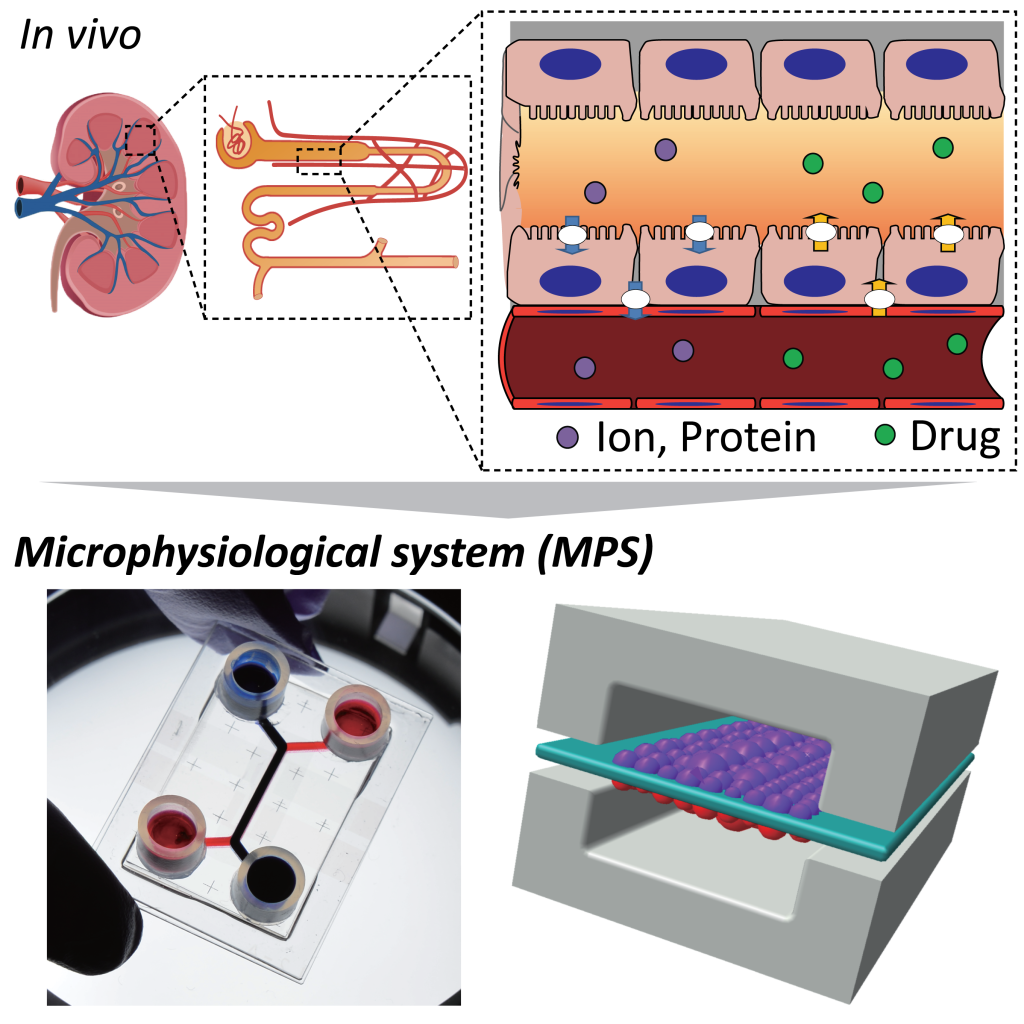
Kidney Proximal Tubule on a Chip (PToC)
The renal proximal tubule is responsible for reabsorption of water, amino acids, and other molecules from the glomerular filtrate which are not meant for excretion. This study focuses on modeling the organ in vitro and quantifying the enhancements in such reabsorption rates using a microphysiological system. We also assess tissue maturation, ion permeability changes and nephrotoxicity with trans-epithelial electrical resistance measurements (TEER).
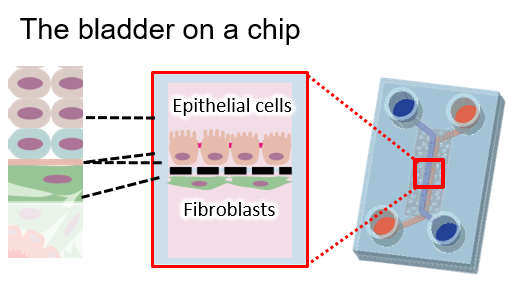
Bladder on a Chip (BoC)
This project involves the modeling the transitional epithelium (multiple layers) of the bladder and quantifying the barrier function on the epithelium. We intend to create a pathological model on this chip that can eventually be used as a platform for drug screening and discovery.
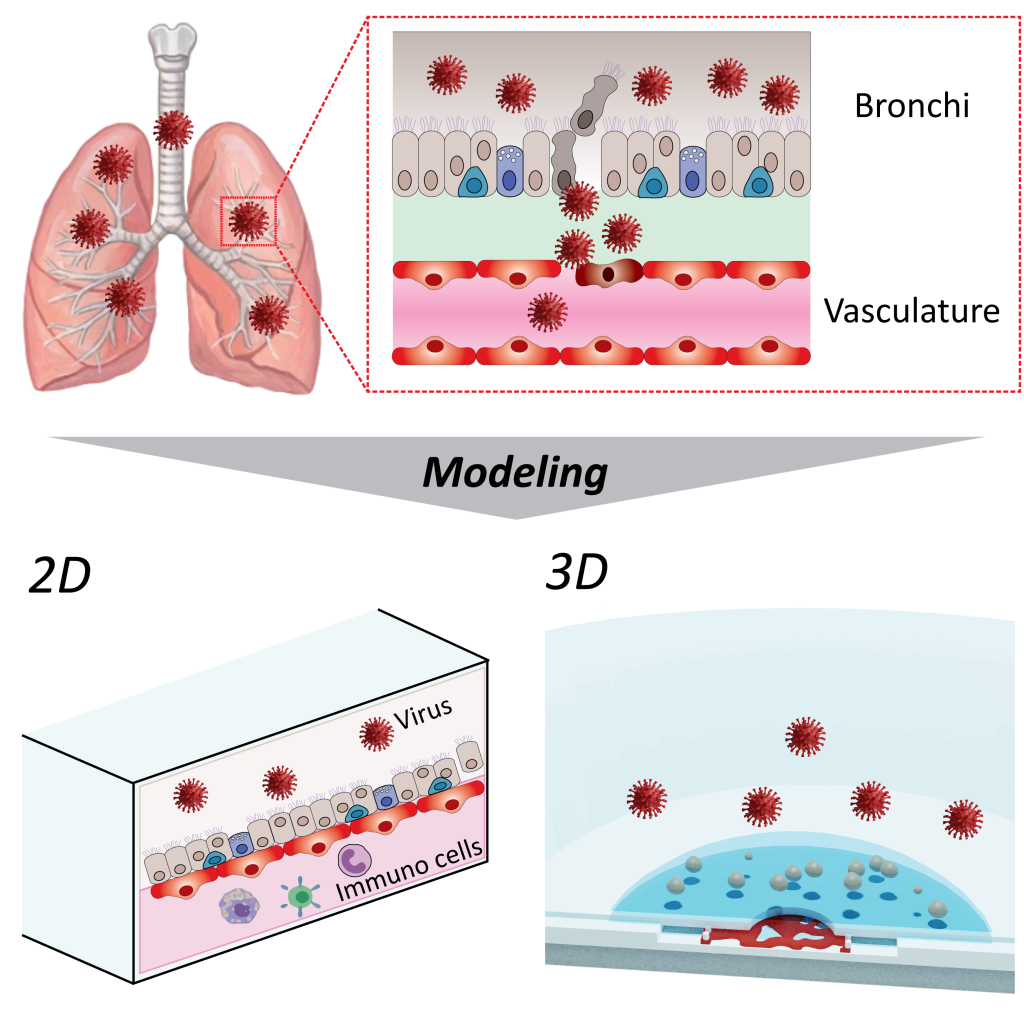
Viral infections
Viral infections such as COVID-19 are caused by infection of various organs, not limited to the nasal cavity and bronchi. Immune response reactions in each organ can affect other organs through the vascular network, causing severe disease. Our laboratory aims to reproduce inflammatory responses in various organs from viral infections using MPS with two-dimensional and three-dimensional vascular network structures.
Application of machine learning in studying vascularized networks
Vasculogenesis, the formation of de novo blood vessels, occurs in several fundamental physiological and pathological processes, including embryonic development, adult angioblast mobilization, and tumor development. Vasculogenesis is accomplished by the morphological changes in endothelial cells. We are trying to analyze the morphological changes of endothelial cells during the vasculogenesis process by using the microphysiological system (MPS) in conjunction with artificial intelligence (AI) visual technology. We are highly interested in the link between their morphology changes and the underlying spatiotemporal gene expressions.

Organoids
Organoids are effective tools for simulating human organ development. However, organoids lack a functional vascular structure, restricting their use in understanding organ development and translational researches. In our group, we established a co-culture system capable of growing organoids with functional three-dimensional vascular system. This model system will additionally be used to better understand the vascularization cues and to screen for early disease markers.